Summary
Cellana tramoserica - common name “true limpet”was discovered by Holten 1802, one of the most diverse and conspicuous herbivore invertebrates on intertidal rocky shores of south-east Queensland. Shells are variable in heights and colours essentially provide protection to the interious body from being exposed in extraordinary environmental conditions with the heat and wave impacts. Many of limpet species exhibit a special behavioural characteristic – “homing” movement which enable to sense and return home after foraging away for feeding in few days. An experiment will be conducted in this project targeting an identification of this behavioural pattern.
Physical Description
C.tramoserica are often living on bare rocks in a high density of community structures. They are exclusively found at the littoral zones as the below (Fig. 1) The Spit, at the Gold Coast Seaway. The geographical feature of The Spit is a deposition landform by accumulative sediments with sand and pebbles. One end is attached to the land and the other end (the nose) projecting into the Gold Coast Seaway.
The edge of nose is built by concrete rocky walls as providing defensive mechanisms over the erosive power from strong waves and winds in the ocean. The rock substrates have subsequently become diverse ecological habitats for vegetated invertebrate and sessile species. C. tramoserica live aggregately on the exposed rocky shores as well as endure the strong wave action, wind speed and tidal cycle effect.
Sexual maturity of C. tramoserica is approximately 24mm in length; adult size can be up to 50-60mm. One of unique identifications is the colourations of radial shell which comprise with yellow, pink or brown, and has ribs in reddish or orange.
Different sizes of limpet are also determined by the structure of radulae. The radular teeth in the large limpets are longer and more widely spaced than the small one. However, the small limpets consider containing a feeding advantage in relation to intraspecific competition due to greater capabilities in grazing food particles within pits on the rock surface (Marshall & Keough,1994).
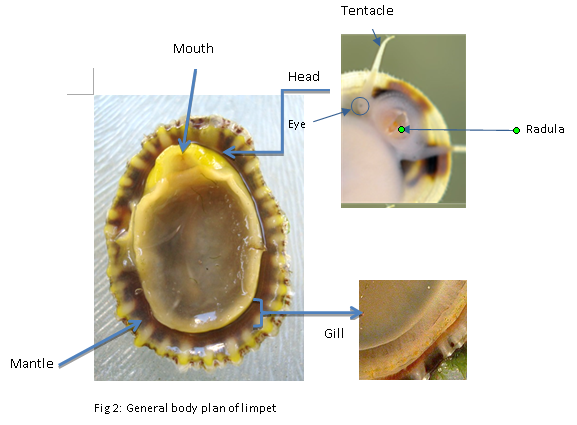
Limpets eat as they go! Mouth is widely opened with radular teeth to search and scrape food off the substrate. The elongated tentacles functioned as detecting surrounding predators (ie. starfish/crabs) whilst feeding. Eyes are located at the base of each tentacle but have less visualising capabilities in term of food search compared with the greater sensibility of the radula (Beatty et al, 2001).
The respiration is through gills underneath the edge of the mantle cavity. Water is drawn under the edge of the mantle and over the gills in order to extract oxygen. The water also associates with flushing waste materials when being passed back out under the shell.

Ecology
The habitat of limpet is considered as harsh and disturbed conditions. The ecosystem of rocky shores is greatly affected by the extreme natural forces from north-east Gold Coast Seaway and the Pacific Ocean. All vegetation and non-vegetated marine wildlife not only require to endure the physical challenges against the strong winds and waves, but also handle chemical stress from highly exposure of UV, salinity and nutrient deficiency in sandy sediments.
On the other hand, the hydrological movement in water provides regulate and stabilise inshore sediment which fundamentally support vegetation developments and binding of sandy particles. Some biological compositions remained existed in the ecosystem as performing important shade, roosting and nursery process for marine life. The habitat niche for common fauna and flora include seaweed, snail, barnacles, marine flatworms and anemones (Fig 3).

Life History & Behaviour
Feeding
C. tramoserica is an opportunistic forager; feeding only when conditions are suitable and preferably being submerged under the tide (Gray& Williams, 2010). They hardly refuge on the rocks with macroalgal growth due to difficulty of adhering on the substrates. The animal gazes to the film of microalgae on the rock surface; remains algal spores and microalgae in cracks and crevices where they are unlikely to reach (Underwood & Jernakoff, 1981).
Reproduction
C. tramoserica is a sexual reproduction with eggs and sperm being broadcast into the sea. Spawning usually occurs in summer season but also correlated with the site. There are less spawned populations from January to March at the middle intertidal but more in low and high intertidal zone (Fletcher 1984). From June to September, larval stages developed over this time when food availability becomes abundant (Fletcher, 1984). Juvenile growth is variable between age groups, but on average the size increased from 6 to 32 mm in about two and a half years. Sexual maturity is reached at an age of one year, when shells are 24 mm in length (Parry, 1982).
Locomotion Experiment
Some limpet species have unique locomotory activity as they recognise their ‘home’ and return to the same spot after the feeding cycle from distance by a recognition of mucus tracks secreted on the rock (Underwood, 1977). Some species move randomly after days of feeding and end up away from the starting point as a new home (Mackay & Underwood, 1977). The various homing behaviours are believably regulated by local density and dispersion to maximise utilisation of the algal food resource.
A design experiment is conducted to further establish whether the rhythms of locomotor exhibited by C. tramoserica on the shore is caused by particular foraging patterns, tide changes or other stimulations from the environmental fluxes such as the thermal effect.
Method
Two groups of 5 specimens were randomly collected for experimental and control test during the low tide (0.8m) of the shore. They were carefully removed by a knife and brought back for experimental setups. The first group was placed in a 30 x 19 x 17(cm) aquarium tank and fed by microalgae attached on the rocks originate collected from the field. The tank was placed in semi-outdoor area so that the photosynthetic process of algae could proceed. The water was also replaced twice a day to maintain the quality of biomass and bacteria available to the limpets and other animals. The limpets also underwent the tidal change by regularly removed and added seawater in the tank twice a day and sprayed the water closely on the shells as pretending to be awashed by the wave action (Gray & Williams, 2010). The control specimens were placed in the same experimental setups without the tidal effect so that they will be submerged in the water throughout the experimental process. The movement pattern of limpets was measured by counting the number of limpets that had any distance changes within 1 hour of the tidal rised and fell in five days. All the specimens, animals and rocks were returned to the original area after completion of the experiment.
The null hypothesis predicts the mean of two groups in their locomotory patterns is the same. The alternative hypothesis considers a significant difference in locomotory responese between the tidal and non-tidal effects. A significant value of P is <0.05. The statistical method was as non-parametric Mann-Whitney one-tailed t test.
Result
The result of hypothesis test in fig.1 indicated as 0.024 which was less than P value <0.05. It concluded that a significant difference in the moving pattern between tidal and non-tidal effect.
Fig 4: Mann-Whitney hypothesis result in SPSS. Significant t-test result = 0.024
|
Test Statistics
|
|
Limpet
|
|
Mann-Whitney U
|
2.000
|
|
Z
|
-2.278
|
|
Exact Sig. (2-tailed)
|
.048
|
|
Exact Sig. (1-tailed)
|
.024
|
|
Point Probability
|
.024
|
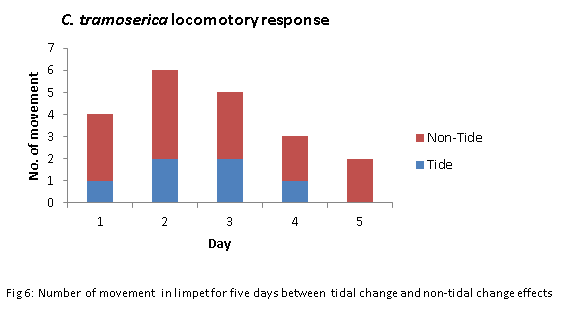
In addition, the mean rank in fig 2 shown that the limpets with tidal impacts was twice lower than the limpets with non-tidal impacts (3.4 <7.6). It indicated a less movement being occurred in the cause of the tidal change compared with the non-tidal change environment. The most active limpets in both two groups were in day 2, followed by day 3. The only movement occurred in day 5 was the non-tidal change group (fig. 6).
Fig 5: the mean rank comparison between Group 1 (3.40) and Group 2 (7.6)
|
Ranks
|
|
Group
|
N
|
Mean Rank
|
Sum of Ranks
|
|
Limpet
|
1
|
5
|
3.40
|
17.00
|
|
2
|
5
|
7.60
|
38.00
|
|
Total
|
10
|
|
|
Group1=Tidal effects Group 2=Non Tidal effects
Discussion
Foraging Behaviour
Factors causing of locomotory response in C. tramoserica are various. The rhythmic foraging behaviour involved maximising the grazing opportunity when feeding competition occurred due to density increased in the populations (Branch & Branch 1980). An observation from the second group found that two closed refuge limpets had moved apart after two days of the experiment. The film of algae on the rocks was also reduced throughout the 5 days experiment which assumed some grazing activities taking place regardless the tidal changes to affect their locomotory responses.
Wave Action
The factor of wave actions causing aggregated movements by the tidal simulation is also predicted. From the first group, 3 out of 5 limpets moved up to the tank wall and remained intact for three days where no tidal was reached. A reason of this is because unfavourable environmental conditions can be avoided by achieving in group actions than individuals (Coleman et al, 2004). It is suggested that this aggregation moving pattern prone to water retention on the surface of rock and airflow between the limpets compared with solitary individuals Coleman (2010). The rising seawater allows the limpets to take water in the mantle to prevent from desiccation.
Thermal Stress
Thermal stress varies in the intertidal zone over multiple spatial and latitudinal scales (Morley et al, 2012) considerably affect the locomotion pattern between emersion and immersion through each tidal cycle. A temperate limpet C. tramoserica have lowest thermal safety margin (TSM) compared with the tropical species such as C.radiate, which presumably would be less physical response required during the tidal change. This considerbly indicated from the group 1 result where the limpets required less movement and water submerge required. However, the thermal factor is not a scope of this experiment as no related data to be collected. Therefore, the further statistical analysis in proving the contribution of locomotion based on thermal stress is needed.
The grazing movement of C tramoserica (video): http://youtu.be/dfJ-IX6njMk
Anatomy & Physiology
The most morphological advantage of C. tramoserica is its disc shaped and muscular foot. It provides ‘super suction’ mechanism to release and adhesion forces of up to 25N contained with its weight less than 25g (Bandyopadhyay et al, 2008). In comparison to human body weight = 70kg only has 9.8N per 1000g. On the dry and hot substratum, it also secretes sticky mucus and seals the bubble water to prevent from drying out.

Evolution & Systematics
Limpets are belonged to the class of Gastropoda. Their significant evolutionary process among gastropod shells is which the coiling characteristic greatly reduced in them. This results a conical shell and a large aperture of ventral part of their body (Denny 2000). This shell shape has achieved several evolution advantages including a better response to thermal and desiccation stress in the intertidal zone; reducing oxygen availability with low-spired shells for a larger gill, and minimising hydrodynamic forces by a large aperture for adhesion the rock (Denny 2000).
The life history of C. tramoserica considers close related to three other species in term of population dynamics (Parry1982). These species are included: Notoacmea petterdi, Patella peroni and Patelloida alticostata (figure 7, 8, 9). They can be found on the same shore near San Remo Melbourne, Victoria in different tidal zone. In the comparison of growth, mortality and reproduction, P. peroni had similar rates of growth and mortality with C.tramoserica but different reproductive effort. In contrast, N. petterdi and P alticostata have the similar reproductive effort with C. tramoserica (Parry 1982).
Biogeographic distribution
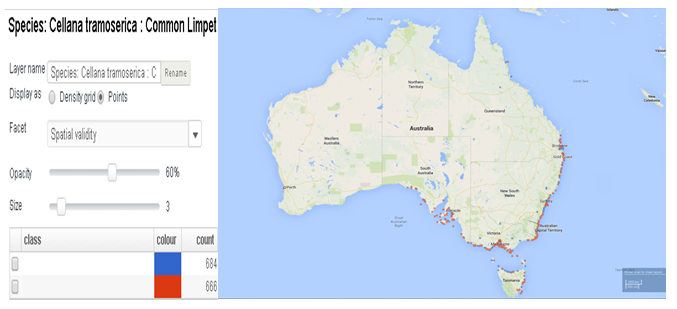
C.tramoserica are distributed national wide in Australia including southernQueensland, New South Wales , Victoria and eastern South Australia, further south in north-east coast of Tasmania.
Conservation & Threats
One of greatest threats of C. tramoserica like any other related molluscs species is human exploitation relating to fossicking and collecting. The removal of large limpets causes a reduction of reproductive rate and genetic discontinuity (Casu et al, 2011). The population becomes extinct when the recovery rate remains low due to absence of effective gene flow from reproductions or immigration.
A protective zone should be identified as prioritising conservation strategies in the low density areas (Casu et al, 2011). The legislation of preserving the population by setting up the rules for collecting and fossicking on the rocky shores should be enforced by the Government sectors. These zones are to allow the increase of genetically differences so that low population can gradually obtained genetically connection with closed related species in extent conservative network (Casu et al, 2011).
References & Links
Bandyopadhyay P. R., Hrubes J. D., Leinhos H. A. 2008. Biorobotic adhesion in water using suctioncups. Biomim3. 1-11.
Beatty R., Bright D., Green J., Kinchen J., MacDonald R.A., Rohr S., Siewerda P.L.,Steffani S., Stewart R., Stickney R., Riddle L.G., Ward B. (2001). Aquatic Life of the World. Marshall Cavendish Corporation, Tarrytown New York.
Branch G.M. and Branch M.L. 1980. Competition between Cellana tramoserica(Sowerby) (gastropoda) and Patiriella exigua (Lamarck) (asteroidea), and their influence on algal standing stocks. Journal of Experimental Marine Biologyand Ecolog., vol 48 ( 1). 35-49.
Casu M., Rivera-Ingraham G. A., Cossu P., Lai T.,Sanna D., Dedola G.L., Sussarellu R.,Gabriella Sella G., Cristo B., Curini-Galletti M., Garcı´a-Go´mez J. C.,Espinosa F. 2011. Patterns of spatial genetic structuring in the endangered limpet Patella ferruginea: implications for the conservation of a Mediterranean endemic Genetica. vol139.1293–1308
Coleman R.A. 2010.Limpet aggregation does not alter desiccation in the limpet Cellana tramoserica. vol386 (1–2). 113–118.
Denny,M.W.2000. Limits to optimization: Fluid dynamics, adhesive strength and the evolution of shape in limpet shells. Journal of experimental biology. vol203(17).2603 – 2622.
Fletcher W.J. 1984. Variability in the reproductive effortof the limpet, Cellanatramoserica.Oecologia. vol 61.259-264.
Gray D. R. G., Williams A. 2010. Knowing when to stop:Rhythms of locomotor activity in the high-shore limpet, Cellana grata Gould. Journal of Experimental Marine Biology and Ecology.vol 391 (1–2, 15). 125–130.
Morley S.A.,Martin S.M., Day R.W., Ericson J., LaiC., Lamare M., Tan K., Thorne M., PeckL.S. 2012. Thermal Reaction Norms and the Scale of Temperature Variation:Latitudinal Vulnerability of Intertidal Nacellid Limpets to Climate Change. PLOSONE. vol7 ( 12).1-8.
Parry G.D.1982.Reproductive Effort in Four Species of Intertidal Limpets. Marine Biology vol67.267-282.
ParryG.D. 1982. The evolution of the life histories of four species of intertidal limpets. Ecological Monographs. vol52(1). 65-91.
Underwood A.J.1977.Movements of intertidal gastropods. Journal of Experimental Marine Biology and Ecology. Vol26(2). 191-201.
Underwood A. J. and Jernakoff P. 1981. Effects of Interactions between Algae and Grazing Gastropods on the Structure of a Low-Shore Intertidal Algal Community. Oecologia.vol 48. 221-233.
|